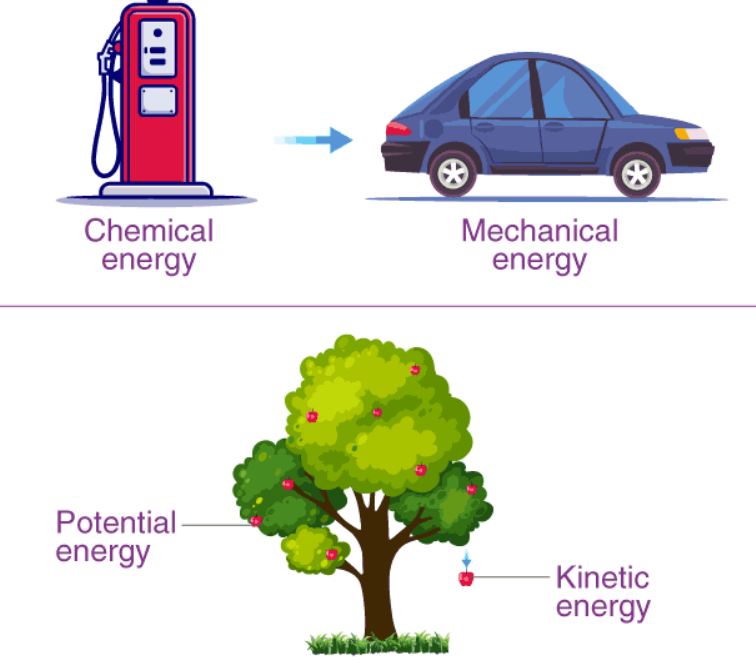
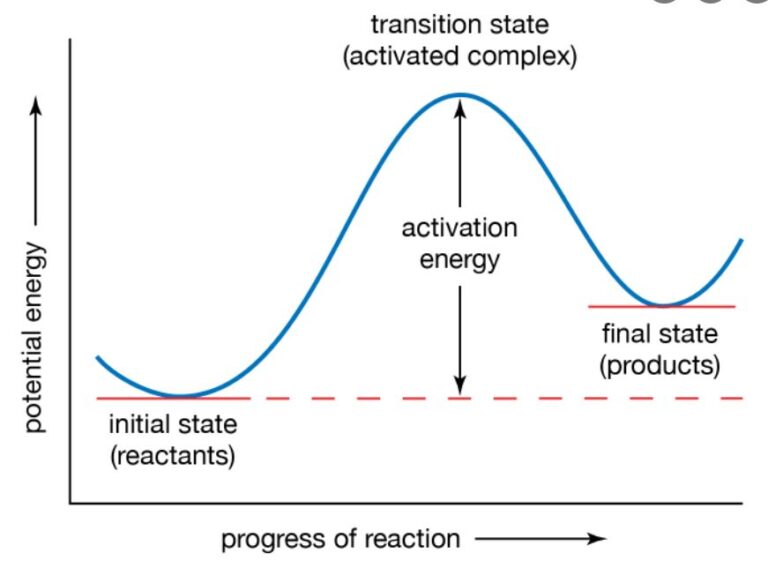
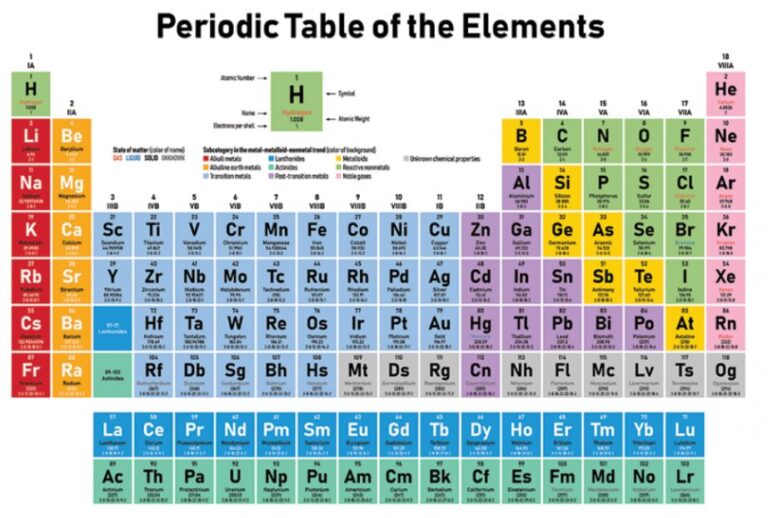
Frequently Asked Kids Chemistry Questions



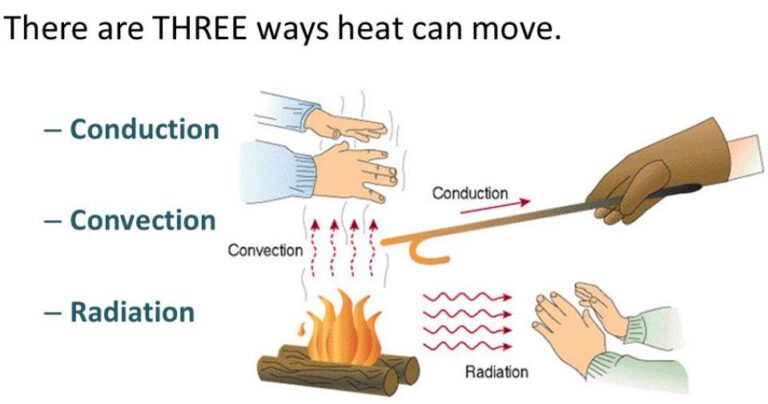
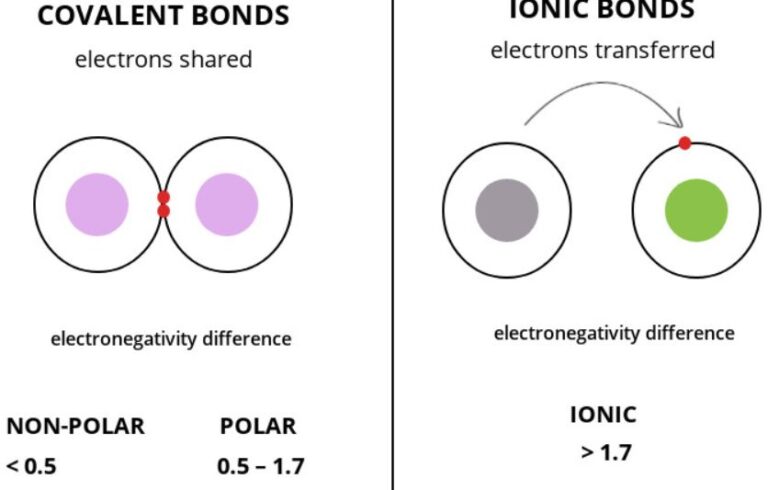
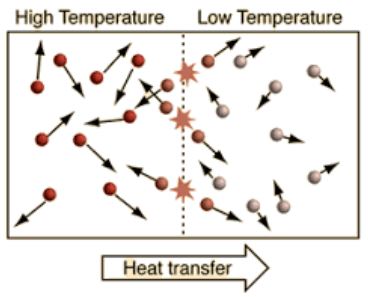
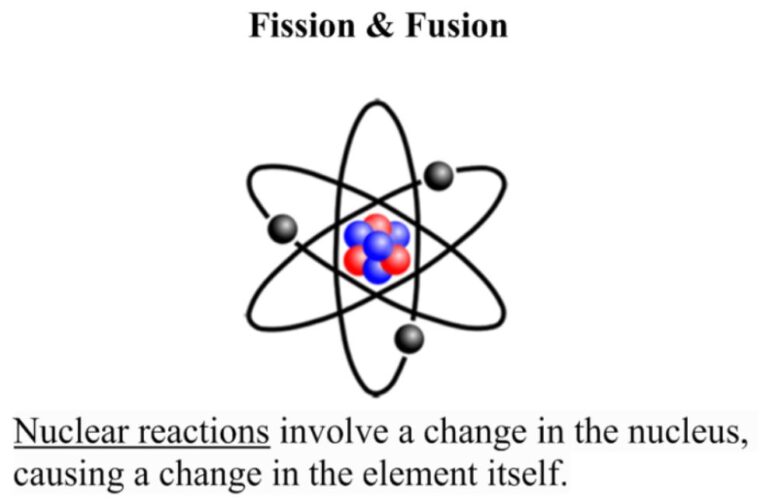
Here is a list of questions and answers about chemistry that kids may ask: What is chemistry? Chemistry is the study of matter and its properties, how it changes, and the energy involved in those changes. What is an atom?…